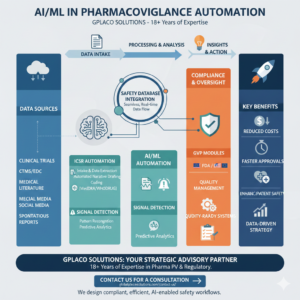
The Strategic Shift: AI/ML in Pharmacovigilance Automation
In the current 2026 regulatory landscape, the implementation of AI/ML in Pharmacovigilance Automation has transitioned from a theoretical concept to a critical operational requirement. Pharmaceutical companies and CROs are facing an unprecedented surge in adverse event volumes, necessitating smarter, faster, and more scalable solutions.
At GPLACO Solutions, we leverage over 18 years of specialized expertise to help organizations navigate this digital transformation, ensuring that the integration of Artificial Intelligence and Machine Learning (ML) aligns perfectly with global safety standards.
The Power of Safety Database Integration
The backbone of any modern PV system is the safety database. However, the true potential of AI/ML in Pharmacovigilance Automation is only realized when these databases are no longer “silos.” Strategic integration allows for a seamless, real-time flow of data, reducing the risk of human error and ensuring that critical safety information is never missed.
Our consulting services focus on connecting your core safety platforms (such as Oracle Argus or ArisGlobal) with:
Clinical Trial Management Systems (CTMS): For unified oversight of site-level safety.
Electronic Data Capture (EDC): To automate the intake of adverse event data from clinical trial sites directly into the safety workflow.
Streamlining Case Processing and ICSR Intake
One of the primary pain points for MAHs and Sponsors is the manual effort required for Individual Case Safety Report (ICSR) processing. By applying AI/ML in Pharmacovigilance Automation, we can automate:
Automated Intake & Extraction: Using Natural Language Processing (NLP) to pull data from unstructured sources like medical literature and physician notes.
Narrative Generation: Leveraging Generative AI to draft clinical narratives that are consistent, accurate, and ready for medical review.
MedDRA Coding: Intelligent auto-coding of terms to ensure global consistency across studies.
Enhancing Signal Detection and Predictive Analytics
Moving beyond simple disproportionality scores, AI/ML in Pharmacovigilance Automation enables advanced Pharmacovigilance Signal Detection. Machine learning algorithms can identify complex patterns across massive datasets—including social media, electronic health records (EHR), and clinical data—to spot potential safety risks much earlier than traditional methods.
With our 18+ years of experience, we don’t just implement technology; we refine the Safety Governance around it. This ensures that your signal management process is proactive, data-driven, and fully compliant with EMA GVP Modules and US FDA requirements.
The Role of GPLACO Solutions in Your AI Journey
Implementing AI is not a “set and forget” task. It requires a deep understanding of Quality Management and Regulatory approvals.
Our advisory services cover:
System Validation: Ensuring that your AI-enabled tools are validated under GVP and 21 CFR Part 11 standards.
Vendor Oversight: Helping you select and audit AI technology vendors to ensure they meet your specific safety needs.
Process Optimization: Redesigning your SOPs to support a “Human-in-the-loop” model, where AI handles the volume and humans handle the complex medical decisions.
Why 18+ Years of Expertise Matters
Technology alone is not a solution. The nuance of drug safety—understanding causality, seriousness, and expectedness—requires a level of clinical judgment that only comes from decades in the field. GPLACO Solutions bridges the gap between sophisticated data science and traditional pharmacovigilance expertise. We ensure that your shift toward AI/ML in Pharmacovigilance Automation results in faster Regulatory approvals and, most importantly, enhanced patient safety.
Conclusion: Future-Proofing Your Safety Governance
The future of pharmacovigilance is automated, integrated, and intelligent. By investing in AI/ML in Pharmacovigilance Automation today, your organization is not just saving costs—it is building a resilient infrastructure capable of navigating the complexities of tomorrow’s global clinical trials.
FAQ: AI/ML in Drug Safety
How does AI reduce pharmacovigilance costs? By automating repetitive tasks like data entry and literature screening, AI reduces manual labor by up to 50%, allowing teams to focus on high-value medical analysis.
Is AI-generated data accepted by the FDA and EMA? Yes, provided the systems are fully validated and a human-in-the-loop oversight process is documented within your Quality Management system.
Can AI help with CTMS and EDC integration? Absolutely. AI-driven middleware can map data between disparate clinical and safety systems, ensuring real-time synchronization.


