FDA Pharmacovigilance Inspection Readiness Checklist | Expert Audit Preparation for 21 CFR 314 & 600

FDA Pharmacovigilance Inspection Readiness Checklist | Expert Audit Preparation for 21 CFR 314 & 600 GPLACO Solutions provides expert consulting and advisory for FDA Pharmacovigilance inspection readiness references related to GVP (Good Pharmacovigilance Practices) audits, it’s important to understand that the U.S. Food and Drug Administration does not use the term “GVP” (that terminology is […]
Signal Management Consulting for FDA and EMA: Early Signal Identification Best Practices

Signal Management Consulting for FDA and EMA: Early Signal Identification Best Practices – Why Signal Management Fails Most failures occur due to documentation gaps, not missed signals. – FDA & EMA Signal Expectations Proactive detection Documented rationale Traceability from data to decision – GPLACO Solutions provides expert Signal Management Consulting Services and implements the best […]
Early-Stage Biotech Pharmacovigilance Consulting for FDA and EMA Compliance
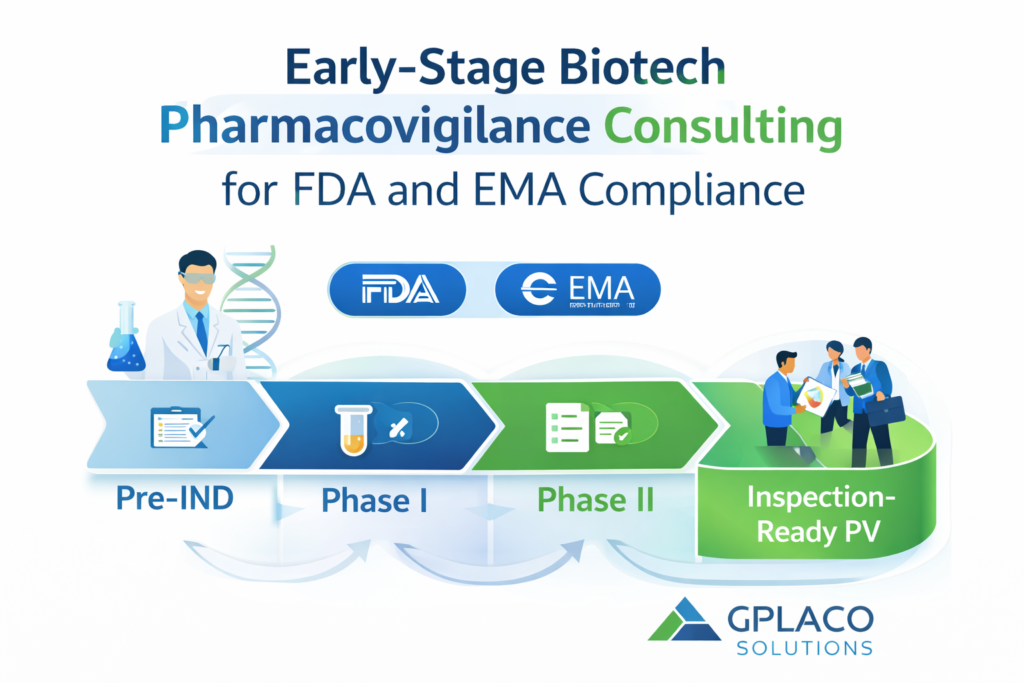
Early-Stage Biotech Pharmacovigilance Consulting: Services for FDA and EMA Compliance Early-stage biotech Pharmacovigilance Consulting is crucial for pharma and biotech companies as they face a unique challenge: regulatory expectations are high, but internal resources are limited. Pharmacovigilance (PV) obligations begin far earlier than many founders expect — often before first-in-human dosing. Many early-stage biotech VPs […]
FDA and EMA Inspection Readiness Consulting: Top Services for GVP Audit and Compliance Checklist

FDA and EMA Inspection Readiness Consulting: Top services for GVP Audit and Compliance Checklist Inspection readiness means being audit-ready every day, not reacting when inspection letters arrive. – What Inspectors Actually Review ICSR quality and timeliness Signal management documentation SOP effectiveness QPPV oversight Vendor governance – Common FDA & EMA Findings Late or inaccurate ICSRs […]
DSUR in Pharmacovigilance: FDA, EMA and ICH E2F Development Safety Update Report Guide

What Is a DSUR in Pharmacovigilance? The DSUR in Pharmacovigilance is Development Safety Update Report (DSUR) is an annual safety report required for investigational products under clinical development. It provides regulators with a global, integrated safety evaluation of a drug, allowing FDA, EMA, and other authorities to assess ongoing benefit–risk. Unlike periodic post-marketing reports, DSURs […]
Pharmacovigilance Outsourcing Services: How and when for Global Pharma, Biotech, and CROs

Pharmacovigilance Outsourcing Services: How and when Global Pharma, Biotech, and CROs Strengthen Safety, Compliance, and Regulatory Success In today’s highly regulated environment, pharmacovigilance outsourcing services have become a strategic necessity for pharmaceutical companies, biotech organizations, sponsors, and CROs operating across the US, EU, UK, and APAC markets. As safety data volumes increase and global regulatory […]
GVP Pharmacovigilance Audits & Due Diligence for MAHs, Sponsors and CROs

GVP Pharmacovigilance Audits & Due Diligence for MAHs, Sponsors and CROs Why Vendor Qualification, PV QMS, and Pre-Outsourcing Readiness Decide Regulatory Outcomes Who this is for: A Marketing Authorisation Holder (MAH) A Sponsor outsourcing pharmacovigilance A Biotech scaling globally Preparing for FDA, EMA, MHRA, TGA inspections 👉 Engage GPLACO SOLUTIONS for risk-based GVP audits and […]
Risk Management Plans Pharmacovigilance (RMPs) : Regulatory Success for the US & EU

Risk Management Plans Pharmacovigilance (RMPs): Proactive Risk Control for Regulatory Success The Risk Management Plans Pharmacovigilance (RMP) is the most critical strategic document in your regulatory dossier. While a Clinical Study Report tells the story of what happened in the past, the RMP is your promise to the FDA and EMA about how you will […]
Expert Signal Detection and Management Services: For US, UK & EU by GPLACO

Expert Signal Detection and Management Services: For US, UK & EU by GPLACO Biggest Industry Challenge Signal Management fails not because signals are missed, but because: Signals are identified too late Statistical outputs are not medically contextualized Documentation is not inspection-ready Signal actions are not traceable end-to-end Health Authorities increasingly expect proactive signal governance, not […]
ICSR Compliance and Quality in Global Pharmacovigilance for the US, EU, UK and MHRA

ICSR Compliance and Quality in Global Pharmacovigilance for the US, EU, UK and MHRA: Building Inspection-Ready Case Processing in Global Pharmacovigilance ICSR compliance and periodic/aggregate reporting is the backbone of global pharmacovigilance and the most frequent source of regulatory findings during inspections. Health Authorities do not assess pharmacovigilance maturity by dashboards or promises — they assess […]


