FDA and EMA Inspection Readiness Consulting: Top Services for GVP Audit and Compliance Checklist

FDA and EMA Inspection Readiness Consulting: Top services for GVP Audit and Compliance Checklist Inspection readiness means being audit-ready every day, not reacting when inspection letters arrive. – What Inspectors Actually Review ICSR quality and timeliness Signal management documentation SOP effectiveness QPPV oversight Vendor governance – Common FDA & EMA Findings Late or inaccurate ICSRs […]
GVP Pharmacovigilance Audits & Due Diligence for MAHs, Sponsors and CROs

GVP Pharmacovigilance Audits & Due Diligence for MAHs, Sponsors and CROs Why Vendor Qualification, PV QMS, and Pre-Outsourcing Readiness Decide Regulatory Outcomes Who this is for: A Marketing Authorisation Holder (MAH) A Sponsor outsourcing pharmacovigilance A Biotech scaling globally Preparing for FDA, EMA, MHRA, TGA inspections 👉 Engage GPLACO SOLUTIONS for risk-based GVP audits and […]
Pharmacovigilance Signal Detection & Management – Training session
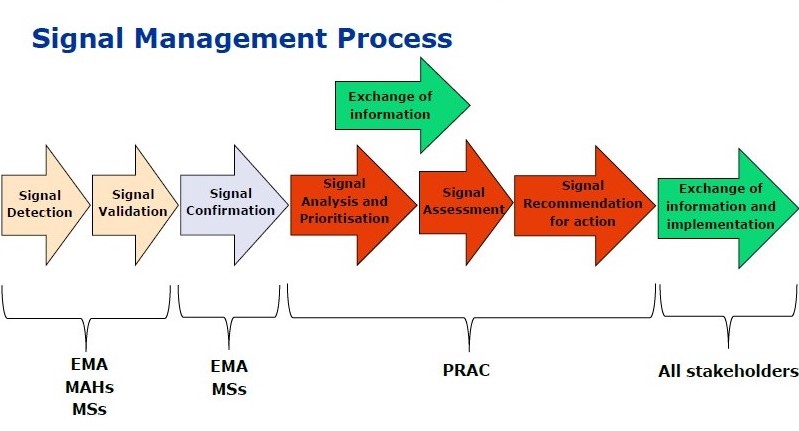
The Essentials of Pharmacovigilance Signal Detection – Pharmacovigilance Signal Detection & Management Training session Effective Pharmacovigilance Signal Detection is critical for patient safety and remains the primary method for identifying previously unknown adverse reactions. During our recent face-to-face session in Bengaluru, we explored how modern data science is transforming this field. The Signal Management Lifecycle […]
Pharmacovigilance System Master File (PSMF) Compliance Guide

Managing the Pharmacovigilance System Master File (PSMF) A Pharmacovigilance System Master File is the most critical document for any MAH operating in the EU and other strictly regulated regions. It provides a detailed description of the pharmacovigilance system used by the marketing authorisation holder with respect to one or more authorised medicinal products. Why the […]
Risk Management Plan in Pharmacovigilance – A Guide

Developing a Risk Management Plan in Pharmacovigilance A Risk Management Plan Pharmacovigilance (RMP) is a document that describes the safety profile of a medicine and the measures used to prevent or minimize risks to patients. Whether you are submitting to the US FDA (as a REMS) or the EMA, the RMP is a mandatory part […]
Periodic Safety update report – PSUR : Submission & Drafting

Best Practices for the Periodic Safety Update Report The Periodic Safety Update Report (PSUR) is a critical tool for post-marketing safety evaluation. It provides an update on the worldwide safety experience of a medicinal product at defined time points after its authorization. The Purpose of a Periodic Safety Update Report The main objective of […]
Periodic Benefit Risk Evaluation Report (PBRER) Requirements

Navigating the Periodic Benefit Risk Evaluation Report The Periodic Benefit Risk Evaluation Report (PBRER) is the international standard for aggregate safety reporting under ICH E2C (R2) guidelines. It represents a shift from simple safety reporting to a more holistic benefit-risk assessment. Evolution from PSUR to PBRER While many still use the terms interchangeably, the Periodic […]
Developmental Safety Update Report (DSUR) – Why and when to submit?

Mastering the Developmental Safety Update Report (DSUR) The Developmental Safety Update Report (DSUR) is the standard for safety reporting during the clinical trial phase of drug development. Its purpose is to provide a comprehensive annual review and evaluation of relevant safety information collected during the reporting period. When to Submit a Developmental Safety Update Report […]
Pharmacovigilance Signal Detection Training: Interview & Expert Guide

Comprehensive Pharmacovigilance Signal Detection Training Whether you are preparing for a professional career or optimizing your company’s safety governance, our Pharmacovigilance Signal Detection Training provides the technical depth required to succeed. Signal detection is the backbone of post-marketing surveillance, ensuring that new safety risks are identified and managed effectively. What You Will Learn This Pharmacovigilance […]
Overcoming Key Pharmacovigilance Outsourcing Challenges

Navigating Pharmacovigilance Outsourcing Challenges While outsourcing safety activities can be cost-effective, it introduces unique Pharmacovigilance Outsourcing Challenges. The primary risk is the loss of direct oversight, which can compromise Quality management and lead to non-compliance. Key Challenges to Monitor SDEA Ambiguity: Poorly defined Safety Data Exchange Agreements. Communication Gaps: Delays in transferring data between the […]


